The best professional team
Our business boasts top of the line services
Branding
Metus vulputate eu felis imperdiet proin nulla aliquet enim tortor at.
Management
Metus vulputate eu felis imperdiet proin nulla aliquet enim tortor at.
Design
Metus vulputate eu felis imperdiet proin nulla aliquet enim tortor at.
Delivery
Metus vulputate eu felis imperdiet proin nulla aliquet enim tortor at.




-
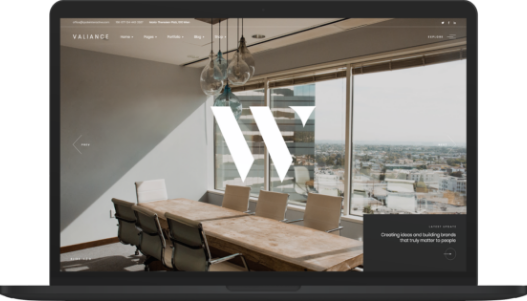
The best prognosis
The economic world and it's development today
Neque volutpat ac tincidunt vitae semper quis lectus nulla at. Nibh nisl id venenatis a condimentum. Libero justo laoreet sit amet. Nisl nisi scelerisque eu ultrices vitae. Dolor sit amet consectetur adipiscing elit pellentesque. Phasellus egestas tellus rutrum tellus pellentesque eu. Sagittis purus sit amet volutpat. -
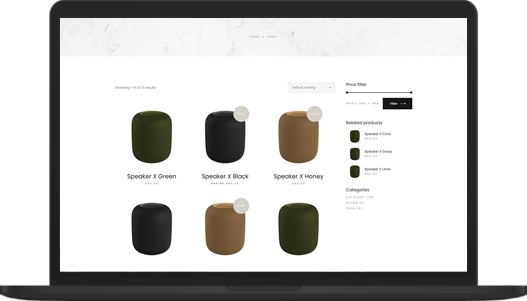
Top economic boost
Great business models of the near future
Neque volutpat ac tincidunt vitae semper quis lectus nulla at. Nibh nisl id venenatis a condimentum. Libero justo laoreet sit amet. Nisl nisi scelerisque eu ultrices vitae. Dolor sit amet consectetur adipiscing elit pellentesque. Phasellus egestas tellus rutrum tellus pellentesque eu. Sagittis purus sit amet volutpat. -

Step in the right direction
The Best Way to Boost Your economic Potential
Neque volutpat ac tincidunt vitae semper quis lectus nulla at. Nibh nisl id venenatis a condimentum. Libero justo laoreet sit amet. Nisl nisi scelerisque eu ultrices vitae. Dolor sit amet consectetur adipiscing elit pellentesque. Phasellus egestas tellus rutrum tellus pellentesque eu. Sagittis purus sit amet volutpat. -
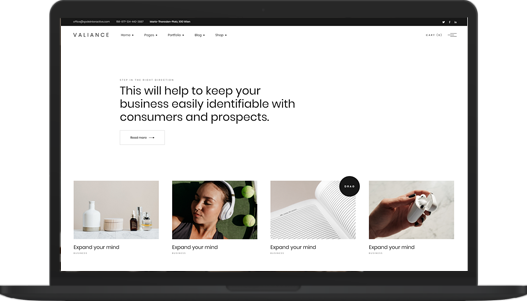
PROACTIVE SPACE
Learn to achieve more with your nine to five
Neque volutpat ac tincidunt vitae semper quis lectus nulla at. Nibh nisl id venenatis a condimentum. Libero justo laoreet sit amet. Nisl nisi scelerisque eu ultrices vitae. Dolor sit amet consectetur adipiscing elit pellentesque. Phasellus egestas tellus rutrum tellus pellentesque eu. Sagittis purus sit amet volutpat.
Our Business
Day-to-day Work Made Simple and easy
Branding
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus.
Manage
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus.
Design
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus.
step in the right direction
Don't be shy, please don't hesitate to say hi
Error: Contact form not found.


